Achondroplasia Market is expected to reach US$ 366.85 Million by 2033 | DataM Intelligence
The Global Achondroplasia Market is expected to reach at a CAGR of 9.9% during the forecast period 2025-2033.
The Achondroplasia Market is witnessing strong growth driven by innovative targeted therapies, rising rare disease awareness, and expanding biologics pipelines, accelerating global treatment adoption.”
AUSTIN, TX, UNITED STATES, February 26, 2026 /EINPresswire.com/ -- Market Overview:— DataM Intelligence
The Achondroplasia Market is the most common form of genetic dwarfism caused by mutations in the fibroblast growth factor receptor 3 (FGFR3) gene, leading to impaired bone growth and skeletal abnormalities. The Achondroplasia Market has gained substantial momentum in recent years due to advancements in precision medicine, increasing awareness regarding rare genetic disorders, and expanding access to early diagnostic technologies. The growing focus on disease-modifying therapies rather than symptomatic management is transforming treatment approaches worldwide, encouraging pharmaceutical companies to invest heavily in innovative biologics and targeted therapies.
To Download Sample Report Here: https://www.datamintelligence.com/download-sample/achondroplasia-market
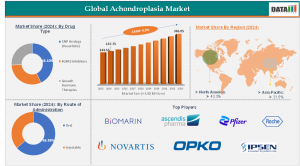
According to DataM Intelligence, The Global Achondroplasia Market was valued at approximately USD 157.75 million in 2024 and is projected to reach nearly USD 366.85 million by 2033, growing at a compound annual growth rate (CAGR) of around 9.9% during the forecast period. Rising approvals of novel therapeutics, supportive government policies for orphan drug development, and increasing newborn genetic screening programs are major contributors to market expansion. The drug therapy segment currently dominates the market due to the emergence of growth-promoting biologics, while North America leads geographically owing to strong healthcare infrastructure, advanced research capabilities, and favorable reimbursement systems supporting rare disease treatments.
The market’s growth trajectory is further strengthened by collaborative research initiatives between biotechnology firms and academic institutions aiming to improve long-term patient outcomes. Increasing patient advocacy efforts and regulatory incentives such as orphan drug designation continue to accelerate innovation, positioning achondroplasia treatment as one of the fastest-evolving segments within the rare disease therapeutics landscape.
Key Highlights from the Report:
Increasing adoption of targeted biologic therapies is reshaping achondroplasia treatment strategies globally.
Rising prevalence of genetic skeletal disorders is driving long-term market demand.
Drug therapy remains the leading segment due to clinical effectiveness and regulatory approvals.
North America dominates the Achondroplasia Market supported by strong reimbursement frameworks.
Expanding pediatric treatment programs are accelerating early intervention adoption.
Strategic partnerships and pipeline innovations are strengthening competitive market dynamics.
Market Segmentation:
The Achondroplasia Market is segmented primarily based on treatment type, route of administration, distribution channel, and end-user. Among treatment types, pharmaceutical therapies represent the most significant revenue contributor. Disease-modifying drugs designed to regulate bone growth pathways are increasingly replacing conventional supportive treatments such as orthopedic surgeries and physical therapy. These innovative therapies directly target underlying genetic mechanisms, improving growth outcomes and quality of life for patients.
Based on the route of administration, injectable therapies dominate due to the biological nature of newly approved medications. Injectable formulations ensure controlled dosing and higher therapeutic efficacy, especially in pediatric populations where treatment consistency is critical. Oral treatment options remain limited but are under development, indicating potential diversification in future treatment modalities.
From an end-user perspective, hospitals and specialty clinics account for the largest share of market utilization. These healthcare settings provide multidisciplinary care involving endocrinologists, genetic specialists, orthopedic surgeons, and rehabilitation experts. Pediatric specialty centers are witnessing increasing patient inflow as early diagnosis becomes more common through neonatal genetic screening initiatives.
Distribution channels include hospital pharmacies, specialty pharmacies, and online pharmaceutical platforms. Specialty pharmacies are emerging as a crucial segment due to the need for temperature-controlled biologics distribution and patient-specific therapy management programs.
Speak to Our Analyst and Get Customization in the report as per your requirements: https://www.datamintelligence.com/customize/achondroplasia-market
Regional Insights:
North America holds the largest share in the Global Achondroplasia Market due to early adoption of innovative therapies and strong regulatory support for orphan drug development. The presence of leading biotechnology companies, well-established research institutions, and advanced healthcare reimbursement models contributes significantly to regional dominance. Additionally, growing patient awareness and structured rare disease registries enable improved treatment accessibility across the United States and Canada.
Europe represents the second-largest market, supported by government-funded healthcare systems and expanding rare disease initiatives. Countries such as Germany, France, and the United Kingdom are investing in genetic research programs and pediatric healthcare improvements, which promote early intervention therapies for achondroplasia patients.
The Asia-Pacific region is expected to witness the fastest growth during the forecast period. Increasing healthcare expenditure, improving diagnostic infrastructure, and rising awareness regarding genetic disorders are driving market penetration in countries like China, Japan, and India. Expanding pharmaceutical collaborations and improving access to advanced biologics are expected to significantly enhance treatment availability across emerging economies.
Latin America and the Middle East & Africa regions are gradually evolving markets, primarily driven by improving healthcare infrastructure and increasing government initiatives supporting rare disease management programs.
Market Dynamics:
Market Drivers
One of the primary drivers of the Achondroplasia Market is the rapid advancement in molecular biology and genetic therapeutics. Breakthrough drug approvals targeting FGFR3 signaling pathways have fundamentally changed treatment outcomes. Increasing investment in rare disease research, combined with orphan drug incentives offered by regulatory agencies, encourages pharmaceutical companies to accelerate product development. Furthermore, rising global awareness among healthcare professionals and parents regarding early diagnosis has significantly improved treatment adoption rates.
Market Restraints
Despite promising growth prospects, the market faces several challenges. High treatment costs associated with biologic therapies remain a significant barrier, particularly in low- and middle-income countries. Limited patient populations also pose commercialization challenges for manufacturers, impacting pricing strategies and reimbursement approvals. Additionally, long-term safety data requirements for pediatric therapies may delay regulatory approvals and market entry of pipeline products.
Market Opportunities
The Achondroplasia Market presents strong opportunities through emerging gene therapy research and personalized medicine approaches. Advancements in genome-editing technologies hold potential for long-term corrective treatments rather than lifelong therapy management. Expanding healthcare access in developing regions and growing telemedicine adoption for rare disease monitoring further create untapped growth avenues. Strategic collaborations between biotechnology firms and research organizations are expected to accelerate innovation and broaden treatment accessibility globally.
Looking For Full Report? Get it Here: https://www.datamintelligence.com/buy-now-page?report=achondroplasia-market
Frequently Asked Questions (FAQs):
How big is the Global Achondroplasia Market currently?
What is the projected growth rate of the Achondroplasia Market through 2033?
Who are the key players operating in the global Achondroplasia treatment market?
What factors are driving growth in the Achondroplasia Market worldwide?
Which region is expected to dominate the Achondroplasia Market during the forecast period?
Company Insights:
Key players operating in the Achondroplasia Market include:
BioMarin Pharmaceutical Inc
Ascendis Pharma A/S
Pfizer Inc.
Roche Holding AG
Novartis AG
Ipsen S.A.
OPKO Health Inc
QED Therapeutics
LG Chem Life Sciences
Recent Developments:
United States:
February 2026: BioMarin initiated a post-marketing study for Voxzogo combining it with limb-lengthening surgery protocols, with early findings presented at a pediatric conference.
January 2026: BridgeBio Pharma released Phase 2 extension data for infigratinib, demonstrating sustained safety and efficacy in long-term use for adolescents. A new national rare disease registry initiative included achondroplasia metrics, supporting better real-world evidence collection.
December 2025: Pfizer announced a collaboration with a biotech firm to develop FGFR3-targeted small molecules, aiming to address unmet needs in surgical adjunct therapies. Reimbursement updates from CMS improved access to biologic treatments, boosting market adoption rates.
November 2025: FDA expanded approval for vosoritide (Voxzogo) to younger pediatric patients, enhancing early intervention options based on new Phase 3 data showing improved height velocity. Ascendis Pharma reported positive interim results from the forward trial for TransCon CNP, positioning it as a potential once-weekly alternative therapy.
Japan:
February 2026: Ascendis Pharma submitted an early data package for TransCon CNP to PMDA, leveraging global trial results with Japan subgroup analysis.
January 2026: Market reports highlighted 15% YoY growth in treatment uptake, fueled by MHLW reimbursement reforms for rare skeletal disorders. Kyowa Kirin dosed first patients in a Japan-specific Phase 3 trial for infigratinib.
December 2025: Approval of an expanded label for vosoritide included combination use with orthopedic interventions, driven by domestic post-approval studies.
November 2025: PMDA granted fast-track designation to Kyowa Kirin's infigratinib program following promising local trial data, accelerating its path to approval. Japan Achondroplasia Society partnered with pharma firms for nationwide genetic screening expansion.
Unlock 360° Market Intelligence with DataM Subscription Services: https://www.datamintelligence.com/reports-subscription
Conclusion:
The Achondroplasia Market is undergoing a transformative phase driven by scientific breakthroughs, increased investment in rare genetic disorders, and growing global awareness. The shift toward disease-modifying therapies marks a significant milestone in improving patient quality of life and long-term health outcomes. With strong pipeline developments, supportive regulatory frameworks, and expanding healthcare accessibility, the market is expected to witness sustained growth over the coming decade. As innovation continues to reshape treatment paradigms, stakeholders across the healthcare ecosystem are positioned to benefit from expanding opportunities within this rapidly evolving rare disease therapeutics sector.
Related Reports:
Aromatase Inhibitor Drug Class Market
Targeted Protein Degradation Market
Sai Kiran
DataM Intelligence 4Market Research
+1 877-441-4866
Sai.k@datamintelligence.com
Visit us on social media:
LinkedIn
X
Legal Disclaimer:
EIN Presswire provides this news content "as is" without warranty of any kind. We do not accept any responsibility or liability for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this article. If you have any complaints or copyright issues related to this article, kindly contact the author above.