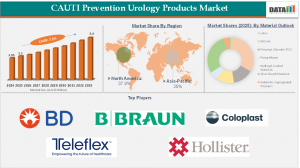
CAUTI Prevention Urology Products Market Growth Projection US$5.9 billion by 2033 Growth in Adoption, Technology (2026)
Top CAUTI Prevention Urology Products Market (2026) | Innovations, Applications & Global Insights
Comprehensive CAUTI Prevention Urology Market | Global Insights & Key Players”
AUSTIN, TX, UNITED STATES, February 26, 2026 /EINPresswire.com/ -- Market Size and Growth (2026)— DataM Intelligence 4Market Research LLP
CAUTI Prevention Urology Products Market was valued at US$3.05 billion in 2024, increased to US$3.3 billion in 2025, and is projected to reach US$5.9 billion by 2033, growing at a CAGR of 7.5% between 2026 and 2033. In response, healthcare facilities are accelerating adoption of advanced prevention solutions such as antimicrobial‑coated urinary catheters, closed drainage systems, catheter securement devices, and comprehensive catheter care kits. The rising prevalence of indwelling catheter usage - especially among elderly and chronically ill patients - coupled with stringent infection‑control regulations, has positioned CAUTI prevention as a critical component of patient safety programs.
Request Executive Sample Report (Get Higher Priority for Corporate Email ID):– https://www.datamintelligence.com/download-sample/cauti-prevention-urology-products-market
Latest M & A
• Teleflex sale of urology business: Teleflex Incorporated agreed to sell its Acute Care and Interventional Urology businesses to Intersurgical Ltd for about $530 million as part of deals also including divestiture of OEM assets, streamlining its portfolio and strengthening the buyer’s urology and related device offering.
Market Drivers & Opportunities
Key factors driving market expansion include:
Increasing Healthcare Expenditure and Patient Safety Focus: Hospitals worldwide are shifting investment toward preventive measures to reduce CAUTI incidence, hospital readmissions, and associated medico‑legal costs. Enhanced infection control strategies improve clinical outcomes and align with quality‑based reimbursement models.
Stricter Regulatory and Reimbursement Policies: Regulatory agencies and accreditation bodies emphasize CAUTI prevention protocols, encouraging adoption of advanced urology products. Hospital infection control committees increasingly mandate the use of antimicrobial and safety‑enhanced catheters to comply with healthcare quality standards.
Technological Innovation: Continuous development in catheter materials, such as antimicrobial, silver‑alloy, and hydrophilic coatings, offers improved bacterial resistance and reduced infection rates, presenting significant deployment opportunities across acute, long‑term, and home care settings.
Growing Elderly and Chronic Care Population: Demographic shifts toward aging populations, increasing rates of urinary incontinence, and chronic urological conditions are driving catheter utilization, thus expanding the demand for prevention products.
Market Segments
By Application Outlook
• Urinary Incontinence Management - Focused on addressing chronic bladder issues and improving patient quality of life.
• Acute Surgical & Perioperative Care - Supports safer catheterization during surgeries and post-operative care.
• Critical Care / ICU - Helps prevent infections in high-risk, catheterized ICU patients.
• BPH & Obstructive Urology - Designed for effective management of benign prostatic hyperplasia and urinary obstruction.
By End-Use Outlook
• Hospitals (Public & Private) - Widely used for procedural safety and infection control.
• Long-Term Care Facilities & Nursing Homes - Essential for geriatric care and chronic catheter management.
• Home Care & Ambulatory Surgical Centers - Supports outpatient procedures and home-based catheter management.
By Material Outlook
• Latex & Silicone Catheters - Preferred for comfort, flexibility, and biocompatibility.
• Silver-Based & Antibiotic-Impregnated - Effective in reducing the risk of CAUTI.
• Hydrogel-Coated & PVC - Improves patient comfort and reduces urethral irritation.
By Technology Outlook
• Antimicrobial & Biofilm-Resistant Surfaces - Critical for infection prevention in high-risk patients.
• Sensor-Enabled / Smart Catheters - Offers real-time monitoring and early infection detection.
• Drug-Eluting & Closed-Loop Drainage - Supports safe post-operative and ICU patient care.
By Patient Type Outlook
• Adult & Geriatric Patients - Primary users due to higher catheterization needs.
• Pediatric Patients - Utilized in specialized urology care centers.
• Long-Term vs Short-Term Users - Catheters designed for both extended use and short-term procedures.
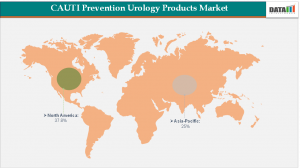
Regional Market Share
• North America: 35%-40% - Dominates market due to advanced printing tech, strong chemical distribution networks, and high industrial usage.
• Japan: 12% - Growing demand from electronics, imaging, and precision chemical industries.
• Europe: 18% - Expansion supported by printing, publishing, and chemical manufacturing adoption.
• Asia-Pacific: 20% - Large-scale industrial, printing, and imaging chemical production in China, India, and Southeast Asia.
• Middle East & Africa: 8% - Emerging adoption in printing and imaging applications with industrial growth.
• Germany: 5% - Steady demand from high-end printing, chemical, and imaging sectors.
• United Kingdom: 4% - Adoption supported by industrial printing and research laboratories.
• South Korea: 3% - Growth from electronics, imaging chemicals, and high-tech manufacturing.
• Canada: 2% - Niche market for chemical imaging and printing applications.
• Spain: 2% - Moderate growth from industrial printing and imaging applications.
Request for Customized Sample Report as per Your Business Requirement:- https://www.datamintelligence.com/customize/cauti-prevention-urology-products-market
Key Developments
✅ February 2026: Hospitals globally accelerated adoption of antimicrobial and silver-coated urinary catheters to reduce catheter-associated urinary tract infections (CAUTIs) and improve patient safety.
✅ January 2026: Manufacturers launched hydrophilic-coated and antibiotic-impregnated catheters, enhancing infection prevention and patient comfort in long-term care and ICU settings.
✅ December 2025: Smart catheter systems with integrated sensors gained traction, enabling real-time monitoring of urine output and early detection of infection risk.
✅ November 2025: Healthcare providers strengthened protocols for CAUTI prevention by adopting evidence-based catheterization guidelines and staff training programs.
✅ October 2025: Rising adoption of single-use and pre-lubricated catheters improved hygiene standards, reduced cross-contamination, and lowered hospital-acquired infection rates.
✅ August 2025: Clinical studies highlighted the efficacy of silver-alloy and coated catheters, driving procurement in urology and critical care departments.
✅ June 2025: Awareness campaigns and hospital-led initiatives increased usage of CAUTI prevention kits,
Key Market Players
The CAUTI prevention urology products market is highly competitive, with global and regional medical device companies innovating and expanding portfolios to address clinical needs. Major participants include:
1. BD (Becton, Dickinson and Company)
2. B. Braun SE
3. Coloplast
4. Teleflex Incorporated
5. Cook Medical
6. Medtronic
7. Hollister Incorporated
8. ConvaTec Group PLC
9. Boston Scientific Corporation
10. Cardinal Health
11. Merit Medical Systems
12. Medline Industries, LP
13. Poiesis Medical LLC
Buy Now & Unlock 360° Market Intelligence:- https://www.datamintelligence.com/buy-now-page?report=cauti-prevention-urology-products-market
Recent Developments - USA
2025 Highlights
Increased Adoption of Antimicrobial‑Coated Catheters: U.S. hospitals are significantly expanding use of antimicrobial‑coated urinary catheters as part of infection‑prevention protocols, in line with tightening CDC infection control guidelines and rising clinical emphasis on CAUTI reduction.
2024 Highlights
Medtronic Launch: Medtronic plc introduced its Harmony™ Spiral Wave™ Reusable Urinary Catheter System in 2024, designed to enhance infection prevention capabilities while reducing environmental impact compared to single‑use devices, supporting sustainability and clinical efficacy.
Recent Developments - Japan
2025 Highlights
Innovation in Catheter Coatings: Japanese healthcare institutions and device manufacturers are advancing silver‑alloy and hydrophilic catheter technologies, improving patient comfort and lowering infection risk, enhancing the overall portfolio of CAUTI prevention products in the region.
2024 Highlights
Industry Endorsements and Market Growth: Japanese urology associations endorsed broader use of antimicrobial catheters in outpatient and long‑term care settings, contributing to notable market growth driven by elderly patient demand and clinical compliance initiatives.
Conclusion
The global CAUTI Prevention Urology Products Market is on a sustained growth trajectory as healthcare systems worldwide prioritize infection prevention, patient safety, and cost‑effective clinical outcomes. With strong technological advancements, supportive regulatory frameworks, and increasing adoption by hospitals and care facilities, the market is well‑positioned to deliver positive impact and substantial economic value through 2033. Continued innovation, strategic partnerships, and region‑specific initiatives will be pivotal in addressing evolving clinical needs and expanding global market reach.
Related Reports
Urology Robotics Devices Market
Interventional Neurology Devices Market
Sai Kiran
DataM Intelligence 4market Research LLP
+1 877-441-4866
sai.k@datamintelligence.com
Visit us on social media:
LinkedIn
YouTube
X
Legal Disclaimer:
EIN Presswire provides this news content "as is" without warranty of any kind. We do not accept any responsibility or liability for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this article. If you have any complaints or copyright issues related to this article, kindly contact the author above.