Biliary Tract Cancers Treatment Market is projected to reaching USD 824.2 million by 2035 at a CAGR of 8.2%.
DE, UNITED STATES, February 26, 2026 /EINPresswire.com/ --
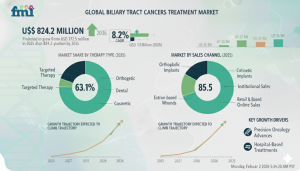
The global Biliary Tract Cancers Treatment Market is expected to grow from USD 372.5 million in 2025 to USD 824.2 million by 2035, expanding at a compound annual growth rate (CAGR) of 8.2%. This growth reflects the rising incidence of cholangiocarcinoma and gallbladder cancer, coupled with increasing awareness, early diagnosis efforts, and major breakthroughs in targeted therapies and immuno-oncology drugs. As these aggressive, often late-detected malignancies see a shift from traditional surgery and chemotherapy toward precision medicine, the market is experiencing accelerated transformation through biomarker-guided regimens and checkpoint inhibitors that deliver better progression-free survival.
Market snapshot: global market 2026 - 2036
Market size in 2026? The Biliary Tract Cancers Treatment Market continues its upward trajectory from the 2025 base of USD 372.5 million, driven by ongoing adoption of novel therapies.
Market size in 2036? While the forecast horizon centers on 2035 at USD 824.2 million, sustained momentum positions the market for continued expansion beyond this period.
CAGR? 8.2% from 2025 to 2035.
Leading product segment(s) and shares — Targeted therapies dominate with a 63.1% share in 2025, fueled by precision approaches to genetic mutations.
Leading material type and share — Not applicable in this oncology context; focus remains on drug classes rather than materials.
Market Momentum (YoY Path)
The Biliary Tract Cancers Treatment Market demonstrates steady progression, building from USD 372.5 million in 2025. Growth accelerates through the late 2020s, with continued expansion evident toward 2028, 2030, and 2031 as targeted therapies and immunotherapies gain broader uptake. By 2033, the market reflects deepening penetration of biomarker-driven options, setting the stage for the 2035 value of USD 824.2 million. Semi-annual updates indicate slightly higher early-decade momentum (H1 2025–2035 at 8.3%) tapering modestly later (H2 at 7.8%), yet overall trajectory remains robust amid regulatory support and pipeline advancements.
Why the Market is Growing
The Biliary Tract Cancers Treatment Market is propelled by the rising incidence of cholangiocarcinoma and gallbladder cancer, enhanced awareness leading to earlier diagnosis, and significant breakthroughs in targeted therapies and immuno-oncology. Emerging options like FGFR2 inhibitors and IDH1 inhibitors show strong efficacy in genetically defined subgroups, especially intrahepatic cholangiocarcinoma cases. The pipeline benefits from trials on PD-1/PD-L1 inhibitors, combination immunotherapies, and tumor microenvironment modulators, expanding choices for refractory patients.
Drivers, Opportunities, Trends, Challenges
Drivers
Immunotherapy is revolutionizing prospects for biliary tract cancers through checkpoint inhibitors (PD-1/PD-L1, CTLA-4) like pembrolizumab and nivolumab, alongside emerging CAR-T approaches. These deliver durable responses in advanced cases. Targeted therapies and precision oncology unlock new frontiers by addressing specific mutations (FGFR2 fusions, IDH1), improving survival with reduced toxicity via agents like pemigatinib and ivosidenib.
Opportunities
Orphan drug designations and regulatory incentives from agencies like the FDA and EMA reduce trial costs, offer tax credits, and grant extended exclusivity (7 years USA, 10 years Europe), alongside fast-track pathways. These lower risks and spur investment from biotechs and pharma in novel therapies for this underserved rare cancer area.
Trends
Precision medicine advances, including genetic profiling and biomarker-guided regimens, drive adoption. Regulatory approvals (e.g., zanidatamab for HER2-positive BTC, durvalumab combinations) and trial momentum expand options. Institutional frameworks support high-cost, monitored therapies.
Challenges
The heterogeneity of biliary tract cancers—varying subtypes, molecular features (FGFR2 fusions, IDH1/KRAS/HER2 alterations)—complicates universal therapy development, patient stratification, trial design, and broad-market applicability. Variable responses and resistance limit duration of benefit, demanding ongoing innovation.
Competitive Landscape
Tier 1 companies lead with 69.3% global share, including AstraZeneca, Incyte, and Merck & Co., Inc., through heavy R&D investment, clinical trials, and strategic alliances. Tier 2 players (30.7% share) such as Daiichi Sankyo Company, Limited, Jazz Pharmaceuticals plc, Relay Therapeutics, Servier Pharmaceuticals LLC, and Taiho Pharmaceutical Co., Ltd. focus on regional influence, partnerships, and agile development. Tier 3 firms like BeiGene, Ltd., Exelixis, Inc., and HUTCHMED target niches. Key players: Incyte, Merck & Co., Inc., AstraZeneca, TAIHO PHARMACEUTICAL CO., LTD, Servier Pharmaceuticals LLC, Jazz Pharmaceuticals plc, Relay Therapeutics, DAIICHI SANKYO COMPANY, LIMITED, BeiGene, Ltd., HUTCHMED, Exelixis, Inc.
Get Access of Report Sample: https://www.futuremarketinsights.com/reports/sample/rep-gb-16764
Scope of the Report
Quantitative units: USD million for value
Segmentation: Drug Classes (Targeted Therapy: FGFR2 inhibitors, IDH1 inhibitor, HER2-targeted agents; Immune Checkpoint Inhibitor: pembrolizumab, durvalumab), Route of Administration (Oral, Intravenous), Line of Therapy (First-line, Second-line), Sales Channels (Institutional Sales, Retail Sales)
Regions: North America, Latin America, Western Europe, Eastern Europe, South Asia and Pacific, East Asia, Middle East and Africa
Countries: United States, Canada, Germany, France, China, Japan, India, South Korea, Brazil, United Kingdom (among others covered)
FAQ
What is the projected size of the Biliary Tract Cancers Treatment Market by 2035? The market is forecasted to reach USD 824.2 million by 2035, growing from USD 372.5 million in 2025.
Which segment leads in the Biliary Tract Cancers Treatment Market? Targeted therapies hold 63.1% share in 2025, while institutional sales dominate distribution at 85.5%.
Which countries show strong growth potential? The UK leads at 9.5% CAGR, followed by Germany (8.7%), Japan (8.6%), USA (8.2%), and China (7.0%), driven by precision medicine, regulatory support, and disease prevalence.
Explore More Related Studies Published by FMI Research:
Japan Biliary Tract Cancers (BTCs) Treatment Market:https://www.futuremarketinsights.com/reports/japan-biliary-tract-cancers-treatment-market
China Biliary Tract Cancers (BTCs) Treatment Market:https://www.futuremarketinsights.com/reports/china-biliary-tract-cancers-treatment-market
Germany Biliary Tract Cancers (BTCs) Treatment Market:https://www.futuremarketinsights.com/reports/germany-biliary-tract-cancers-treatment-market
Australia Biliary Tract Cancers (BTCs) Treatment Market:https://www.futuremarketinsights.com/reports/australia-biliary-tract-cancers-treatment-market
About Future Market Insights (FMI)
Future Market Insights, Inc. (FMI) is an ESOMAR-certified, ISO 9001:2015 market research and consulting organization, trusted by Fortune 500 clients and global enterprises. With operations in the U.S., UK, India, and Dubai, FMI provides data-backed insights and strategic intelligence across 30+ industries and 1200 markets worldwide.
Why FMI: Decisions that Change Outcomes- https://www.futuremarketinsights.com/why-fmi
Contact Us:
Future Market Insights Inc.
Christiana Corporate, 200 Continental Drive,
Suite 401, Newark, Delaware – 19713, USA
T: +1-347-918-3531
Website: https://www.futuremarketinsights.com
Have a specific Requirements and Need Assistant on Report Pricing or Limited Budget please contact us - sales@futuremarketinsights.com
Sudip Saha
Future Market Insights Inc.
+1 347-918-3531
email us here
Legal Disclaimer:
EIN Presswire provides this news content "as is" without warranty of any kind. We do not accept any responsibility or liability for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this article. If you have any complaints or copyright issues related to this article, kindly contact the author above.