Hemophilia Therapeutics Market to Reach US$22.9 Bn by 2033 | Persistence Market Research
The hemophilia therapeutics market is expanding with advancements in treatments, increased awareness, and broader access to innovative therapies worldwide.
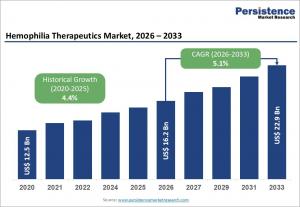
LONDON, UNITED KINGDOM, April 16, 2026 /EINPresswire.com/ -- The global hemophilia therapeutics market is projected to experience significant growth in the coming years, with an estimated market size of US$16.2 billion by 2026. It is expected to reach US$22.9 billion by 2033, growing at a compound annual growth rate (CAGR) of 5.1% during the forecast period from 2026 to 2033. The market's growth is driven by an increasing prevalence of hemophilia A and hemophilia B, better diagnosis rates, and improved disease awareness.
Download Your Free Sample & Explore Key Insights: https://www.persistencemarketresearch.com/samples/4727
Key Drivers of Market Growth
National health systems and patient advocacy organizations are leading efforts to enhance early diagnosis, expanding the treated patient pool. In developed markets, advanced therapies continue to receive reimbursement, supporting premium pricing and consistent revenue growth. Emerging economies are improving healthcare access through public procurement programs and broader insurance coverage, contributing to incremental market volume expansion.
Technological innovations in treatment, including extended half-life clotting factor concentrates and non-factor therapies such as bispecific antibodies, are improving patient adherence and reducing bleeding episodes. Gene therapy platforms are making significant strides through regulatory approvals and are beginning to reshape treatment paradigms, offering the potential for durable factor expression following a single administration.
Key Industry Highlights
Regional Leadership: North America is expected to dominate the market, holding around 48% of the share in 2026, thanks to high healthcare spending and advanced infrastructure. The Asia Pacific market, however, is set to grow the fastest, with a projected CAGR of approximately 6.5% through 2033, driven by expanding healthcare access and awareness.
Dominant Disease Indication: Hemophilia A will lead the market with nearly 74% of the revenue share in 2026. However, Hemophilia B is expected to grow the fastest due to the adoption of gene therapies.
Leading Therapeutic Category: Factor replacement therapies will maintain a dominant position with a projected 61% revenue share in 2026, due to their established efficacy.
Fastest-growing Therapeutic Category: Non-factor and gene therapies are forecasted to grow at a robust CAGR of 6.6% from 2026 to 2033, thanks to their reduced infusion frequency and improved patient outcomes.
Healthcare Setting Trends: Hospitals and hemophilia treatment centers (HTCs) are projected to account for 60% of the market share in 2026, though home care and specialty clinics are expected to grow fastest due to patient convenience and home infusion adoption.
Technological Advancements and Market Potential
The growing burden of hemophilia, combined with advancements in treatment technologies, is reshaping the therapeutic landscape. According to the U.S. Centers for Disease Control and Prevention (CDC), approximately 33,000 males in the U.S. are living with hemophilia, and improvements in early diagnosis have boosted access to care. The World Federation of Hemophilia (WFH) notes that up to 75% of individuals with inherited bleeding disorders remain undiagnosed, which underscores the significant gaps in disease awareness, screening, and access.
Technological advancements such as gene therapies and non-factor biologics are contributing to more durable and effective treatment options, reducing infusion frequency and improving treatment adherence. These innovations are particularly welcomed by patients and healthcare providers who are increasingly seeking therapies that offer better outcomes with reduced treatment burden.
Get Custom Insights Designed for Your Businecss: https://www.persistencemarketresearch.com/request-customization/4727
Market Challenges: High Costs and Regulatory Hurdles
Despite the significant advances in hemophilia treatment, high costs remain a major barrier to widespread adoption. Gene therapies, such as valoctocogene roxaparvovec, can exceed US$2 million per patient, making them prohibitively expensive in many markets, especially in low- and middle-income countries. Limited insurance coverage and out-of-pocket expenses add to the financial burden, slowing adoption rates despite the clinical benefits of these therapies.
Regulatory complexities further complicate market expansion. Gene therapies and biologics require rigorous evaluation by regulatory authorities, which prolongs time-to-market and increases development risks. Additionally, the manufacturing process for these advanced therapies is highly specialized and involves intricate quality control measures, which can result in supply bottlenecks and limit scalability.
Opportunities in Emerging Markets
Emerging markets, especially in Asia Pacific and Latin America, present substantial growth opportunities for hemophilia therapeutics. Improved healthcare infrastructure and rising disease awareness are creating a favorable environment for the adoption of both traditional and advanced therapies. The Asia Pacific region, in particular, is projected to experience the fastest market growth, driven by structural healthcare improvements and a large undiagnosed patient population.
China, for example, approved the first gene therapy for hemophilia B—Dalnacogene Ponparvovec Injection—in 2025, marking a significant milestone for the region. The approval and uptake of gene therapies in these emerging markets are expected to accelerate further as public health programs, awareness campaigns, and specialized training for healthcare providers continue to expand.
Competitive Landscape
The global hemophilia therapeutics market is moderately consolidated, with major players such as Roche, Sanofi, Novo Nordisk, Bayer, and Pfizer commanding a significant portion of the market. These companies leverage their extensive clinical expertise, strong relationships with healthcare providers, and broad therapeutic portfolios, which include factor replacement therapies, non-factor biologics, and gene therapies. Investment in research and development remains a critical focus for these companies to maintain their leadership in the market.
Regional and niche players, including BioMarin, uniQure, and CSL Behring, are also making strides by focusing on specialized therapies for rare bleeding disorders.
Get Custom Insights Designed for Your Businecss: https://www.persistencemarketresearch.com/request-customization/4727
Market Segmentation
By Disease Indication
Hemophilia A
Hemophilia B
Hemophilia C
Others
By Therapeutic Category
Factor Replacement Therapies
Non-Factor Therapies
Gene Therapies
Immune Tolerance Induction (ITI)
Adjunct & Supportive Therapies
By Healthcare Setting
Hospitals & Hemophilia Treatment Centers (HTCs)
Specialty Clinics
Home Care
Retail Pharmacies & E-Commerce Channels
By Region
North America
Europe
East Asia
South Asia & Oceania
Latin America
Middle East & Africa
Read Related Reports:
DNA/RNA Extraction Market: The global DNA/RNA extraction market grows from US$ 1.6 Bn in 2026 to US$ 2.6 Bn by 2033, at a 7.2% CAGR during forecast 2026–2033.
Speech Therapy Service Market: The global speech therapy service market grows from US$ 44.9 Bn in 2026 to US$ 83.1 Bn by 2033, at a 9.2% CAGR during forecast 2026–2033.
Persistence Market Research
Persistence Market Research Pvt Ltd
+1 646-878-6329
email us here
Visit us on social media:
LinkedIn
Instagram
Facebook
YouTube
X
Legal Disclaimer:
EIN Presswire provides this news content "as is" without warranty of any kind. We do not accept any responsibility or liability for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this article. If you have any complaints or copyright issues related to this article, kindly contact the author above.